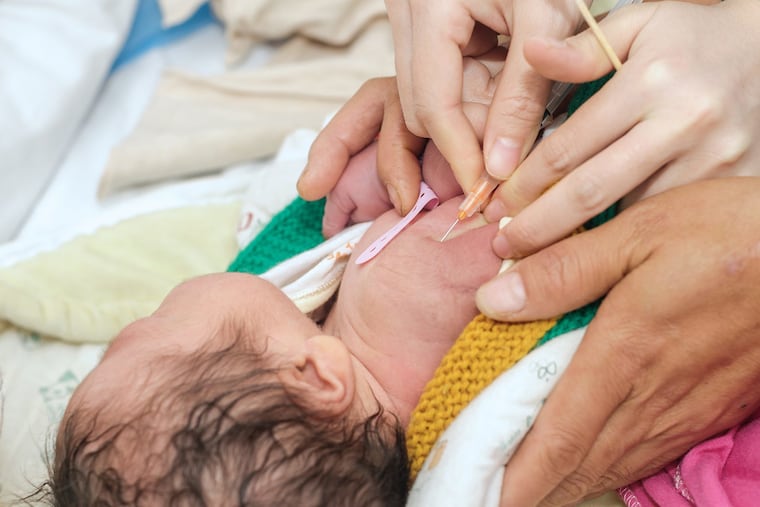
Hepatitis B Vaccine Schedule Under Review: Key Information and Updates on Ongoing Debates
The Advisory Committee on Immunization Practices (ACIP), the foremost vaccine advisory panel in the United States, is set to convene to discuss the potential postponement of the initial dose of the hepatitis B vaccine, a discussion that has sparked considerable debate. Originally developed in Philadelphia, this vaccine has been recommended for all newborns at birth to combat a disease that poses serious long-term health risks, including irreversible liver damage and the absence of a cure.
While the hepatitis B vaccine is widely accepted as both safe and effective, its administration protocol has come under intense scrutiny, particularly from the current administration, which has questioned both the timing and necessity of the birth dose. The upcoming deliberations have been prompted by a confluence of opinions from various stakeholders, including vaccine skeptics added to the ACIP’s ranks during a recent restructuring.
In prior discussions this September, ACIP members deferred a vote on the subject, leading to increased anticipation for the scheduled debate. Advocates for early vaccination argue that delaying the birth dose could expose unvaccinated infants to the dangerously contagious hepatitis B virus, which can be transmitted from infected mothers, as well as through various household sources like personal grooming items contaminated with blood. Public health experts express particular apprehension regarding any possible consultation to restrict vaccination to only those infants whose mothers are confirmed to have the virus, as significant numbers of mothers remain untested.
Supporters of the universal vaccination protocol point to a dramatic decline in hepatitis B infection rates among children and adolescents—reportedly falling by 99% since the introduction of the vaccine in 1991. Before the implementation of universal hepatitis B vaccination at birth, a significant percentage of infections were traced back to maternal transmission.
The discourse surrounding the vaccine comes at a time of heightened awareness of public health infrastructure, with renewed focus on the virus, which remains incurable and can lead to severe liver conditions over time. Leading medical societies maintain that there is no scientific basis for altering the current immunization guidelines. Experts caution that relying on targeted vaccination may create gaps in immunity, particularly given that a notable number of women, between 15 and 16%, still do not undergo testing for hepatitis B during pregnancy.
As these discussions progress, they highlight not only the scientific and medical considerations regarding hepatitis B vaccination but also the implications for public health policy, underscoring the critical need for a balanced approach that prioritizes both safety and efficacy in vaccine administration. This evolving dialogue continues to engage stakeholders across the spectrum, from health professionals to policymakers, reflecting the overarching imperative to protect newborns and ensure widespread immunity against this potentially devastating virus.